Eventually, these individual laws were combined into a single equation—the ideal gas law—that relates gas quantities for gases and is quite accurate for low pressures and moderate temperatures. We will consider the key developments in individual relationships , then put them together in the ideal gas law. The behavior of gases can be described by several laws based on experimental observations of their properties. The pressure of a given amount of gas is directly proportional to its absolute temperature, provided that the volume does not change (Amontons's law).
The volume of a given gas sample is directly proportional to its absolute temperature at constant pressure (Charles's law). The volume of a given amount of gas is inversely proportional to its pressure when temperature is held constant (Boyle's law). Under the same conditions of temperature and pressure, equal volumes of all gases contain the same number of molecules (Avogadro's law).
Gases whose properties of P, V, and T are accurately described by the ideal gas law are said to exhibit ideal behavior or to approximate the traits of an ideal gas. An ideal gas is a hypothetical construct that may be used along with kinetic molecular theory to effectively explain the gas laws as will be described in a later module of this chapter. Although all the calculations presented in this module assume ideal behavior, this assumption is only reasonable for gases under conditions of relatively low pressure and high temperature.
In the final module of this chapter, a modified gas law will be introduced that accounts for the non-ideal behavior observed for many gases at relatively high pressures and low temperatures. A common use of Equation 10.23 is to determine the molar mass of an unknown gas by measuring its density at a known temperature and pressure. This method is particularly useful in identifying a gas that has been produced in a reaction, and it is not difficult to carry out. A flask or glass bulb of known volume is carefully dried, evacuated, sealed, and weighed empty.
It is then filled with a sample of a gas at a known temperature and pressure and reweighed. The difference in mass between the two readings is the mass of the gas. The volume of the flask is usually determined by weighing the flask when empty and when filled with a liquid of known density such as water. The use of density measurements to calculate molar masses is illustrated in Example 10. The ideal gas law allows us to calculate the value of the fourth variable for a gaseous sample if we know the values of any three of the four variables . Some applications are illustrated in the following examples.
The approach used throughout is always to start with the same equation—the ideal gas law—and then determine which quantities are given and which need to be calculated. Let's begin with simple cases in which we are given three of the four parameters needed for a complete physical description of a gaseous sample. Thus the ideal gas law does a good job of approximating the behavior of real gases at STP. Calculations involving the mole can be used to determine unknown concentrations, volumes and masses in reactions.
One mole of any gas occupies 24 cubic decimetres at room temperature and pressure. The most common molar volume is the molar volume of an ideal gas at standard temperature and pressure (273 K and 1.00 atm). The ideal gas law is used like any other gas law, with attention paid to the units and making sure that temperature is expressed in kelvins. However, the ideal gas law does not require a change in the conditions of a gas sample. The ideal gas law implies that if you know any three of the physical properties of a gas, you can calculate the fourth property. 22.Which of the following statements is false?
The properties of N2 will deviate more from ideality at -100oC than at 100oC. Van der Waal's equation corrects for the non-ideality of real gases. Molecules of CH4 at high pressures and low temperatures have no attractive forces between each other. Molecules of an ideal gas are assumed to have no significant volume. Real gases do not always obey the ideal gas laws.
Examples and practice problems of solving equation stoichiometry questions with gases. We calculate moles with 22.4 L at STP, and use molar mass and mole ratios to figure out how many products or reactants we have. It is important to check your answer to be sure that it makes sense, just in case you have accidentally inverted a quantity or multiplied rather than divided. In this case, the temperature of the gas decreases. Because we know that gas volume decreases with decreasing temperature, the final volume must be less than the initial volume, so the answer makes sense. We could have calculated the new volume by plugging all the given numbers into the ideal gas law, but it is generally much easier and faster to focus on only the quantities that change.
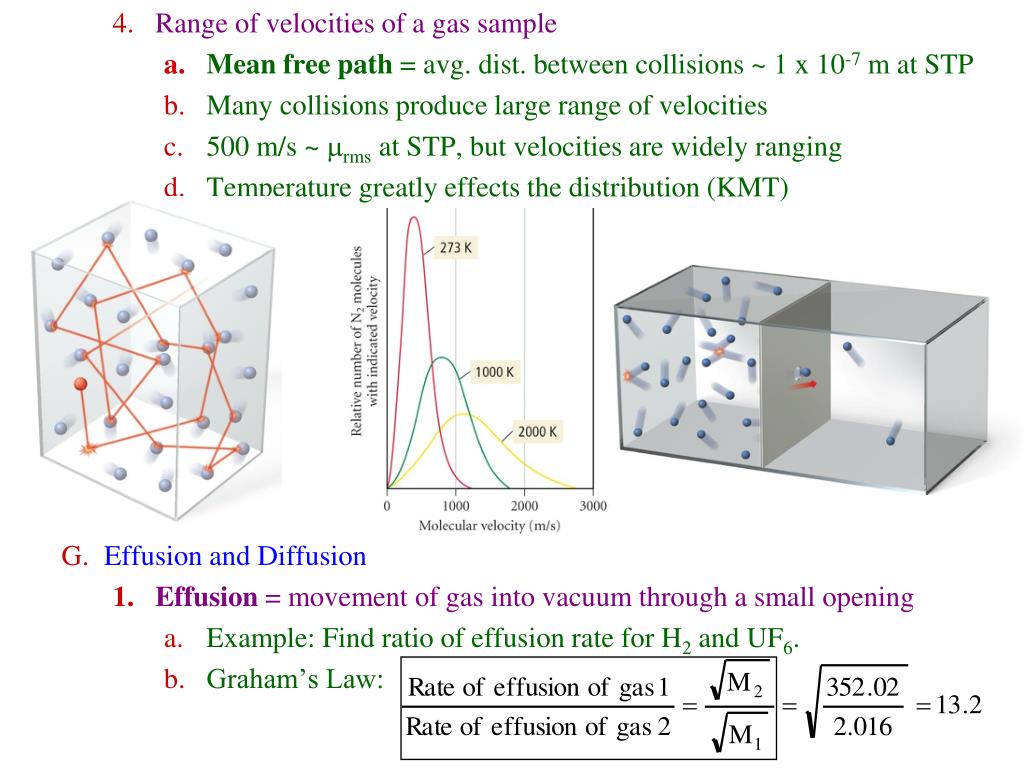
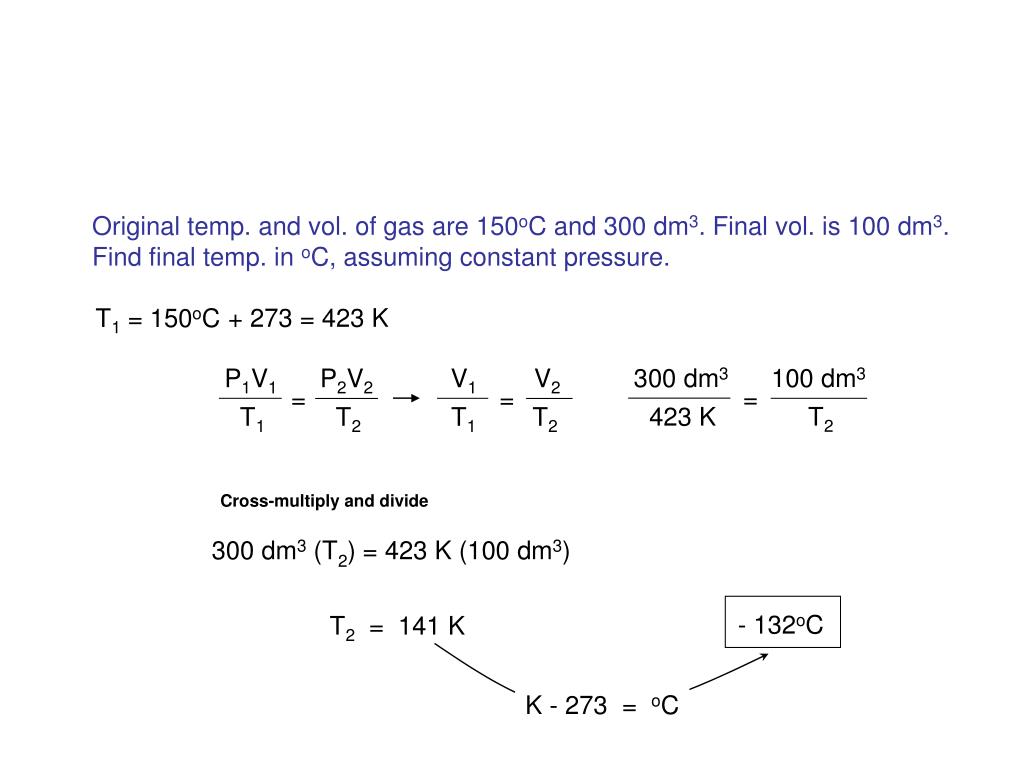
The volume and temperature are linearly related for 1 mole of methane gas at a constant pressure of 1 atm. If the temperature is in kelvin, volume and temperature are directly proportional. Charles's law states that the volume of a given amount of gas is directly proportional to its temperature on the kelvin scale when the pressure is held constant.
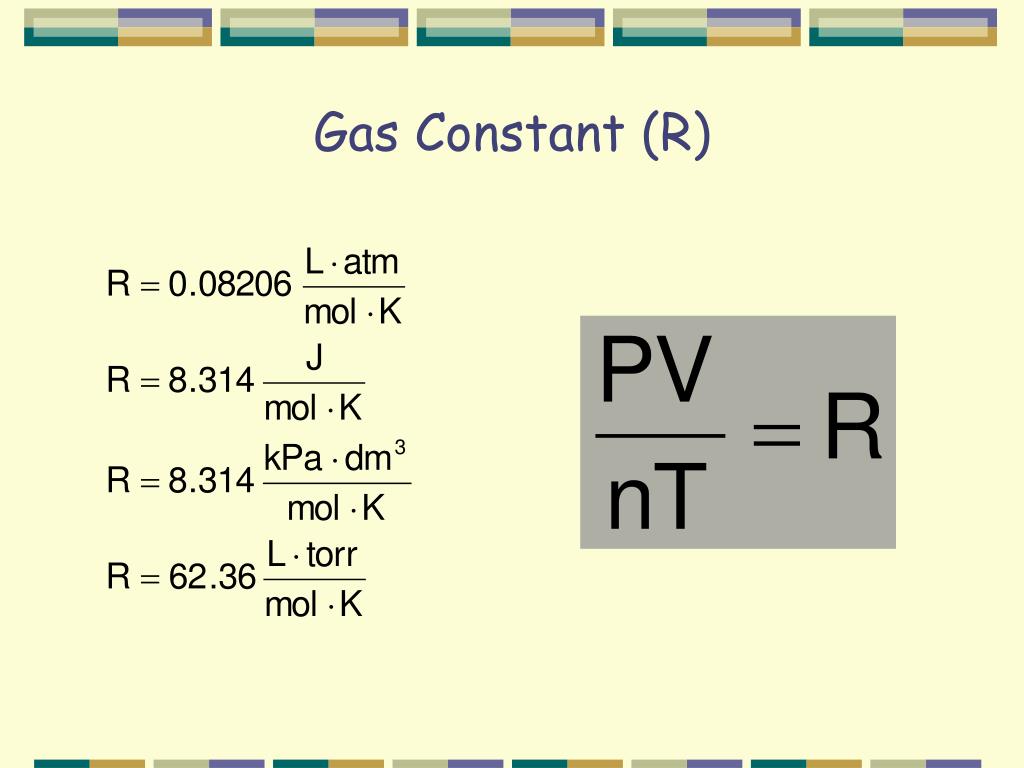
It relates the four independent properties of a gas at any time. The constant R is called the ideal gas law constant. Its value depends on the units used to express pressure and volume.
Table 6.1 "Values of the Ideal Gas Law Constant R" lists the numerical values of R. The reaction of a copper penny with nitric acid results in the formation of a red-brown gaseous compound containing nitrogen and oxygen. A sample of the gas at a pressure of 727 mmHg and a temperature of 18°C weighs 0.289 g in a flask with a volume of 157.0 mL. Calculate the molar mass of the gas and suggest a reasonable chemical formula for the compound. Is defined as a hypothetical gaseous substance whose behavior is independent of attractive and repulsive forces and can be completely described by the ideal gas law. In reality, there is no such thing as an ideal gas, but an ideal gas is a useful conceptual model that allows us to understand how gases respond to changing conditions.
As we shall see, under many conditions, most real gases exhibit behavior that closely approximates that of an ideal gas. The ideal gas law can therefore be used to predict the behavior of real gases under most conditions. ⚗️ Our standard temperature and pressure calculator is a simple tool that can make your life easier! Here you can easily compute both the volume and the number of moles of a gas in standard conditions. 19.Which one of the following statements about the following reaction is false?
CH4 + 2O2 CO2 + 2H2O Every methane molecule that reacts produces two water molecules. If 32.0 g of oxygen reacts with excess methane, the maximum amount of carbon dioxide produced will be 22.0 g. If 11.2 liters of methane react with an excess of oxygen, the volume of carbon dioxide produced at STP is (44/16)(11.2) liters.
If 16.0 g of methane react with 64.0 g of oxygen, the combined masses of the products will be 80.0 g. If 22.4 liters of methane at STP react with 64.0 g of oxygen, 22.4 liters of carbon dioxide at STP can be produced. The ideal gas law can be used in stoichiometry problems in which chemical reactions involve gases. Apply the ideal gas law to molar volumes, density, and stoichiometry problems. The volume of gas produced during a chemical reaction can be measured by collecting the gas in an inverted container filled with water. The gas forces water out of the container, and the volume of liquid displaced is a measure of the volume of gas.
B Make sure that all quantities are given in units that are compatible with the units of the gas constant. If necessary, convert them to the appropriate units, insert them into the equation you have derived, and then calculate the number of moles of hydrogen gas needed. A sample of gas has a mass of 0.570 g . Its volume is 109 mL at a temperature of 95 ∘C and a pressure of 754 mmHg .Find the molar mass of the gas. 15.Which one of the following statements is not consistent with the kinetic-molecular theory of gases? Individual gas molecules are relatively far apart.
The actual volume of the gas molecules themselves is very small compared to the volume occupied by the gas at ordinary temperatures and pressures. The average kinetic energies of different gases are different at the same temperature. There is no net gain or loss of the total kinetic energy in collisions between gas molecules. The theory explains most of the observed behavior of gases at ordinary temperatures and pressures. 1 mole of every gas occupies the same volume, at the same temperature and pressure. The molar volume is the volume occupied by one mole of a substance at a given temperature and pressure.
A sample of methane has a volume of 25 mL at a pressure of 0.86 atm . What is the final volume, in milliliters, of the gas at each of the following pressures, if there is no change in temperature and amount of gas? Part A Calculate the final volume for the sample at 0.31 atm . Express your answer using two significant figures. Part B Calculate the final volume for the sample at 1.90 atm .
Part C Calculate the final volume for the sample at 2200 mmHg . Part D Calculate the final volume for the sample at 69.0 torr . B Convert all known quantities to the appropriate units for the gas constant being used. Substitute the known values into your equation and solve for the molar mass.
Temperature is sometimes measured with a gas thermometer by observing the change in the volume of the gas as the temperature changes at constant pressure. The hydrogen in a particular hydrogen gas thermometer has a volume of 150.0 cm3 when immersed in a mixture of ice and water (0.00 °C). When immersed in boiling liquid ammonia, the volume of the hydrogen, at the same pressure, is 131.7 cm3. Find the temperature of boiling ammonia on the kelvin and Celsius scales. STP in chemistry is the abbreviation for Standard Temperature and Pressure.
STP most commonly is used when performing calculations on gases, such as gas density. The standard temperature is 273 K (0° Celsius or 32° Fahrenheit) and the standard pressure is 1 atm pressure. A Calculate the molar mass of butane and convert all quantities to appropriate units for the value of the gas constant. Imagine filling a rigid container attached to a pressure gauge with gas and then sealing the container so that no gas may escape.
If the container is cooled, the gas inside likewise gets colder and its pressure is observed to decrease. Since the container is rigid and tightly sealed, both the volume and number of moles of gas remain constant. If we heat the sphere, the gas inside gets hotter and the pressure increases. Calculate the molar mass of a gas using the ideal gas law. Intro To Chem - Introduction Chemistry is the science of matter, especially its chemical reactions, but also its composition, structure and properties. Chemistry deals with atoms and their interactions with other atoms, and particularly with the properties of chemical bonds.
Chemistry also involves understanding the properties and interactions of individual atoms and molecules for use in larger-scale applications. Calculate the volume of each gas sample at STP. Express your answer using three significant figures. At the same temperature and pressure equal volumes of all gasses contain the same number of molecules. Use the molar volume of a gas at STP to calculate the density (in g/L ) of carbon dioxide gas at STP.
We calculate moles with the Ideal Gas Law, because the conditions are not at STP, and use molar mass and mole ratios to figure out how many products or reactants we have. Use the ideal gas law to derive an equation that relates the remaining variables for a sample of an ideal gas if the following are held constant. Radon is a radioactive gas formed by the decay of naturally occurring uranium in rocks such as granite.
It tends to collect in the basements of houses and poses a significant health risk if present in indoor air. Many states now require that houses be tested for radon before they are sold. B Rearrange the ideal gas law to isolate those quantities that differ between the initial and final states on one side of the equation, in this case V and T. In this section, we describe how these relationships can be combined to give a general expression that describes the behavior of a gas. The ideal gas equation contains five terms, the gas constant R and the variable properties P, V, n, and T.
How To Find The Volume Of A Gas Sample Specifying any four of these terms will permit use of the ideal gas law to calculate the fifth term as demonstrated in the following example exercises. Assume that the hydrogen gas is collected at a pressure of 1.0 atm and temperature of 319 K Express your answer using two significant figures. Standard temperature and pressure , a.k.a. standard conditions, describe the environment of a chemical reaction.
The density of a gas is constant as long as its temperature remains constant. Gases can be expanded without limit. Gases diffuse into each other and mix almost immediately when put into the same container. The molecular weight of a gaseous compound is a non-variable quantity. Pressure must be exerted on a sample of a gas in order to confine it. Is the volume occupied by one mole of any gas, at room temperature and pressure.
Standard temperature and pressure are a useful set of benchmark conditions to compare other properties of gases. If you have the mass of the gas, you can divide the mass by the molecular weight of the gas molecules to get the number of moles. Then multiply this by 22.4 Liters / mole to get the volume. For example, if you have 96 g of O2, then you can divide by the molecular weight of O2, which is 32 g/mol, to get 3 moles. In Example 5, we were given three of the four parameters needed to describe a gas under a particular set of conditions, and we were asked to calculate the fourth.
We can also use the ideal gas law to calculate the effect of changes in any of the specified conditions on any of the other parameters, as shown in Example 6. The standard conditions calculations are based on the ideal gas law formula. First, let's calculate the volume of our substance in STP temperature and pressure conditions. Calculate volume of ammonia gas produced at STP when 140g of nitrogen gas is reacted with 30g of hydrogen gas. Use the molar volume of a gas at STP to determine the volume occupied by 10.5g of neon at STP.
A gas occupies a volume of 410mL at 27 degrees Celcius and 740mm Hg pressure. Calculate the volume the has would occupy at STP. Each flask has the same number of gas molecules. The velocity of the gas molecules is the same in each flask. The density of each gas is the same. There are twice as many O2 and H2 molecules as Ne atoms.